- Home
- About Us
- Work
- Journal
- Contact
- Intellij ultimate edition price
- Xamarin for visual studio 2010
- Etka 7-4 2015
- Game guardian 6-0-5 apk
- Watermark in fotostation
- Eplan training
- Cd3 profantasy
- Garvo garasiyo gujarati song
- Flowjo 10 normalization histogram
- Mob psycho 100 season 2 episode 9 stream
- Dragon ball hyper dimension quotes
- Malena movie actress name
- Usgs 3d earthquake map
- Home
- About Us
- Work
- Journal
- Contact
- Intellij ultimate edition price
- Xamarin for visual studio 2010
- Etka 7-4 2015
- Game guardian 6-0-5 apk
- Watermark in fotostation
- Eplan training
- Cd3 profantasy
- Garvo garasiyo gujarati song
- Flowjo 10 normalization histogram
- Mob psycho 100 season 2 episode 9 stream
- Dragon ball hyper dimension quotes
- Malena movie actress name
- Usgs 3d earthquake map
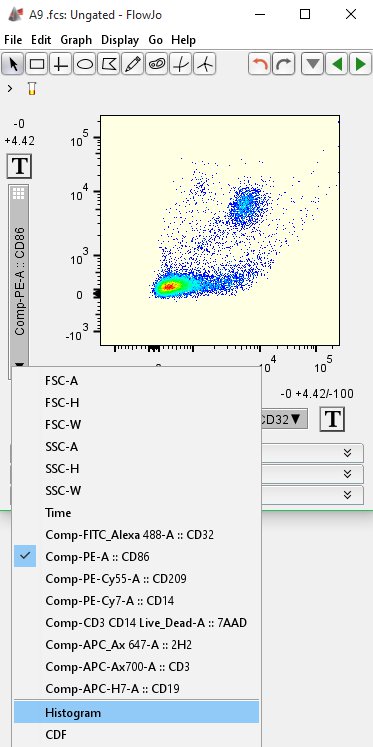
Subsetting gates rely on the expression levels of markers in the analysis, and what those makers identify. This is where the major work of data analysis is done. As a result, approximately 45,000 more events have been eliminated from the analysis. These events represent debris, cell fragments and pyknotic cells. The events with very low FSC and SSC, as well as those with low FSC and high SSC are eliminated. When cells pass through the laser intercept and fluoresce, the photons are converted to an electronic pulse in the photomultiplier tube.Īs shown in the figure below, the major density of events is captured by this gate. This gate is used to remove doublets from the dataset and is particularly useful with digital data. Interestingly, when this program was run on over 29,000 files in the FlowRepository, the authors showed almost 14% had fluorescent anomalies.įailure to address this problem reduces the sensitivity of all experimental measurements and may result in inaccurate data and results. The red gate on the right-handed plot was used to remove the first seconds of the run where the instrument was in the process of stabilizing the run and not yet ‘flowing’ evenly.Ī recent paper published by Fletez-Brant et al., introduced an automated program in R called “flowClean”, which can do this process in an unbiased, automated fashion. Either forward scatter or side scatter are good choices, as they are both intrinsic measurements of all events passing through the laser intercept. The data are plotted against a time parameter versus a scatter parameter. These plots show the sample running evenly over the time of acquisition. An example of this is shown in the below plots. The principle of this step is to ensure a good and even flow stream during the instrument’s run.Ĭlogging, back-pressure and other instrument-related issues can affect the flow, so eliminating cells that may have been affected by such problems is an important step to cleaning up the data.

The details on this hierarchy, including how each fits together sequentially to produce the optimal flow cytometry figure for every experiment, are outlined below… 1. Backgating - to provide visualization of cells in final gate at higher level.This is where Fluorescence Minus One (FMO) controls become critical in defining the populations of interest. Using viability dyes and dump channels further narrow to the cells of interest. Subsetting gating - to rely on expression of markers and what they identify.Forward and side scatter gating - to remove debris and other events of non-interest while preserving cells based on size and or complexity.Pulse geometry gating - to remove doublets from the dataset.Flow stability gating - to capture events once the flow stream has stabilized, eliminating effects of clogging, back-pressure, and other instrument issues.To this end, the following hierarchy was created to help you gate your events correctly… Hopefully, you will objectively choose the right events to display. In other words, you can reuse and refine your gates and plots over and over again without actually losing cells, but you and you alone will determine which events you are displaying.
While actual cells will not be lost in trying various gating strategies, data points can be eliminated from your population. 5 Gating Strategies For Publishing Flow Cytometry Data This is also where science becomes an art form. Thankfully, there are many ways to avoid shaping the results, and instead sifting for the real and actual data that is relevant to the flow cytometry experiment at hand.Ĭommunicating the results of a flow cytometry experiment is where the researcher has the power to make new or subtle findings instantly comprehensible to the audience. Science must be objective, or it is simply an exercise in creative sculpting, which does nothing to move science forward. The critical difference between sculptor and scientist is that while the sculptor is guided by a creative vision, the researcher is guided by very particular laws of nature and a specific method of working through a biological hypothesis to avoid shaping the results to his or her whims. There is a story inside the data, and it is the job of the researcher to unravel it. When sitting down to perform a new analysis of flow cytometry data, it is much like Michelangelo staring at a piece of marble. “Every block of stone has a statue inside it and it is the task of the sculptor to discover it.” - Michelangelo
